We’ve all heard over and over again on not only this blog but many media outlets that wine (particularly red wine) contains certain compounds that render the beverage beneficial for many aspects of human health. There is a growing trend in research and ultimately in commercial use (for some companies) to create a “super wine”, or in other words a wine that not only has the positive health benefits related to the polyphenol composition of the solution, but also lacks some of the more “negative” components such as the alcohol. Note: alcohol has been shown to contribute to the health benefits of wine when consumed in moderation, but is bad for you when consumed in heavy amounts.
Research into reducing alcohol levels in wine have produced several different techniques including those done before, during, and after fermentation. A more promising technique (according to some researchers) for effectively removing
![Photo By Quinn Dombrowski (originally posted to Flickr as Wine) [CC-BY-SA-2.0 (http://creativecommons.org/licenses/by-sa/2.0)], via Wikimedia Commons](http://www.academicwino.com/wp-content/uploads/2013/05/wine_The_Academic_Wino-300x199.jpg)
Photo By Quinn Dombrowski (originally posted to Flickr as Wine) [CC-BY-SA-2.0 (http://creativecommons.org/licenses/by-sa/2.0)], via Wikimedia Commons
Getting a little more technical, OD is what is known as a “membrane separation process” which means that volatile compounds can be removed from a liquid by a membrane that absorbs the specific volatile compounds you’d like to remove. Passage across this membrane is a function of vapor pressure differences between the liquid, the membrane, and the resulting liquid on the other side. In other words, if you have a membrane that is specific for absorbing ethanol, the vapor pressure of the ethanol is greater inside the original solution, therefore simply by the laws of physics and chemistry, the reaction will move from the largest pressure to the smallest pressure, so the ethanol will be absorbed into the attractive membrane and diffused through it to the other side into the new solution that has a lower vapor pressure of ethanol.
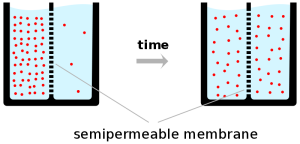
The graphic below illustrates this point a little better than how I just described it: start off with a lot of “stuff” on one side (in our case, the “stuff” is the vapor pressure of the ethanol). The “semipermeable membrane” in our example would be the membrane that specifically absorbs ethanol. Over time, the ethanol will be absorbed into the membrane and transferred to the solution on the other side until an ethanol equilibrium is met (i.e. same amount of ethanol on one side as the other). Once you have an equilibrium, you can start all over again by taking the new solution (which now has less alcohol—note the fewer dots in the figure) and doing the same thing over again. You’ll start with a solution with less ethanol, pass it through the membrane, and end up with a new equilibrium with even less ethanol present. If you’re still confused by this, please ask and I’ll try to clear it up more!
To date, only studies examining the partial removal of alcohol have been performed, and the research on wines completely stripped of alcohol is few and far between. Therefore, the goal of the study presented today was to determine the effect of the complete removal of alcohol from wine using osmotic distillation (OD) on the chemical composition of red wine. The results of this research could help with the movement to create a wine that is rich in the positive cardiovascular and other health benefits without the negative effects of too much alcohol.
Methods
The red wine used in this study was made from the Aglianico grape variety that was grown in the Campania region of Italy in 2009.
For those of you familiar with osmotic distillation: the feed and stripping flow rates used were 70mL/min and 140mL/min, respectively, which according to the authors was chosen based on previous research on the subject.
The OD processed occurred over 5 cycles (i.e. performing the technique I briefly described above 5 times), with the first cycle occurring over 60 minutes, and the remaining cycles occurring over 45 minutes. Again, these times were chosen based on previous research. The removal of alcohol was deemed complete when the alcohol level in wine was less than 0.5%.
The following were measured at the beginning, during, and the end of the experiment for all wine samples: alcohol content, pH, total acidity, phenolic compounds, color intensity, tonality, flavanols, tartaric esters, organic acids, and other aromatic compounds.
Results
• The presence of other compounds in the wine (i.e. aromatic compounds, etc) did not influence the success or rate of the alcohol removal process.
• The alcohol level achieved after 5 cycles of the alcohol removal process was 0.19% alcohol by volume.
• There were no significant differences between any of the wine samples at any alcohol content level in terms of pH, total acidity, total phenolic compounds, flavanols, tartaric esters, and organic acids.
• There were no significant color differences between the original wine and the wine after one cycle of alcohol removal (a loss of 6.5% alcohol by volume).
o The remaining cycles of alcohol removal resulted in a significant increase in color intensity, which according to the authors may be due to the solubility of wine pigments at different alcohol levels.
• After the first alcohol removal cycle, isoamyl alcohol levels significantly decreased (by 66%). Isoamyl alcohols are responsible for some of the aromatic complexity of wine, so this decrease is important.
o Isoamyl alcohols were reduced between 95 and 99% during the remaining alcohol removal cycles.
• Ester concentrations decreased with decreasing levels of alcohol, specifically losing about 10% after the first alcohol removal cycle.
o Ethyl lactate, and monoethyl succinate were unchanged, while ethyl hexanoate, ethyl octanoate, β-phenylethyl acetate, ethyl acetate, and isoamyl acetate were all significantly reduced or lost.
o Total ester loss was about 90% after the 5 alcohol removal cycles.
• After the 5 alcohol removal cycles, acetic acid and hexanoic acid decreased by over 50% of their initial concentrations, while butanoic acid, heptanoic acid, and octanoic acid levels remained unchanged.
• Pantolactone significantly decreased after the first alcohol removal cycle, while 1-hydroxy-2-propanone, phenylacetone, acetoin, and y-butryolactone did not significantly decrease until after the second alcohol removal cycle.
• 2-methoxyphenol was completely removed after the first alcohol removal cycle.
• 2-ethylphenol and acetaldehyde did not significantly decrease until the fourth alcohol removal cycle.
• Sulfur compounds were 80% lower than the phenols and aldehydes in the final wine with complete alcohol removal, which as the author’s state is not a bad thing since sulfur compounds are related to off flavors and aromas in wine!
• Total volatile compounds decreased by 50% after the first alcohol removal cycle, and decreased by 80% after the second alcohol removal cycle.
• Only 1.2% of the original volatile compound concentration remained at the end of the 5th alcohol removal cycle.
Conclusions
The results of this study showed that osmotic distillation successfully removed nearly all of the alcohol from Aglianico red wine. After 5 alcohol removal cycles, the researchers were able to achieve a wine with 0.19% alcohol by volume, with can be considered basically non-alcoholic. Interestingly, the results showed that much of the chemical composition of the wine remained unchanged throughout the alcohol removal process, such as pH, total acidity, total phenolic compounds, flavanols, tartaric esters, and organic acids. However, it was clear from the results that using osmotic distillation to completely remove the alcohol from the wine significantly decreased or completely removed several of the compounds that are responsible for aromatic and flavor characteristics of the finished wine.
From these results, it is clear that more work needs to be done before one can simply use osmotic distillation to create a non-alcoholic wine without disrupting the overall quality of the beverage. According to the authors, coupling osmotic distillation with the supplementation or addition of some of the aromatic compounds lost during the process may result in a finished wine that tastes just like the original but without all the alcohol. In my opinion, I am skeptical about this idea. Simply removing the alcohol while leaving all else constant (say if
those aromatic compounds that were lost during the alcohol removal process were added back in afterward) will still change the overall experience of the finished wine, in my opinion. I’m not talking about the feeling of getting buzzed or sloshed, but the interaction between the alcohols with the other compounds within the wine. Chemistry is a complicated thing, so messing with one part of the recipe is going to change the overall taste or experience, even if you’ve added flavoring back into the mix after it had been lost during the alcohol removal process. Alcohol not only contributes to the flavor part of the story, but it also contributes in a tactile sort of way (in my mouth anyway!). Removing that gives you grape juice, not “non-alcoholic wine”.I understand the desire for some to create a wine-like beverage without the alcohol for certain health reasons, however, a lot more research needs to be done on the different techniques to achieve this goal, and whether or not all these manipulations are financially or otherwise logical in terms of overall cost-benefit ratios are concerned. Why spend a lot of time and money creating a “non-alcoholic wine” from an original alcoholic wine when you can just make grape juice, which has many of the same health benefits that you’re going for in the non-alcoholic beverage?
I’d be curious to see more research on this topic, in addition to some sensory analyses (which were lacking in this study). How do these non-alcoholic “wines” taste after this process? Can you easily supplement the wine with the aromatic compounds that were lost during this process to create a similar tasting beverage as the original alcoholic wine? How does the non-alcoholic wine with or without aromatic supplementation compare to straight up grape juice?
What do you all think of this study (and others like it?). What other questions does this study raise for you? What would you like to see next? Please feel free to leave your comments!
Source: Liguori, L., Russo, P., Albanese, D., and Di Matteo, M. 2013. Evolution of quality parameters during red wine dealcoholization by osmotic distillation. Food Chemistry 140:68-75.

![Photo By Paolo Neo [Public domain], via Wikimedia Commons](http://www.academicwino.com/wp-content/uploads/2013/05/red_wine_The_Academic_Wino-300x225.jpg)
4 comments for “Creating a Non-Alcoholic Wine by Osmotic Distillation: Are Quality Characteristics Retained During the Alcohol Removal Process?”